fluorescence
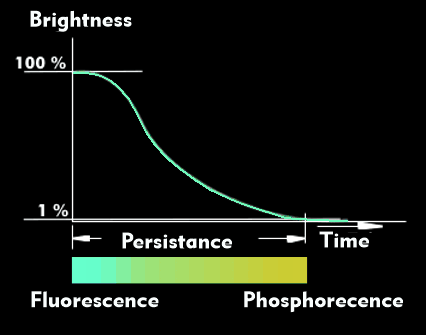
Fluorescence is the first short-lived light emission produced when light-emitting materials are excited. In the case of phosphorus, fluorescence transitions to phosphorescence after decay.
In fluorescence, the emitting material is activated with light whereby the electrons from the conduction band fall back into the valence band and occupy the vacancies. This produces emission in the form of electromagnetic radiation or light emission.
The extremely short-lived fluorescence, which can be only a few nanoseconds or microseconds long, has a different wavelength than the excitation light and also differs in the emitted color temperature from the subsequent phosphorescence, the afterglow, as known from phosphors, for example. In contrast to fluorescence, phosphorescence can last from a few milliseconds to several minutes.