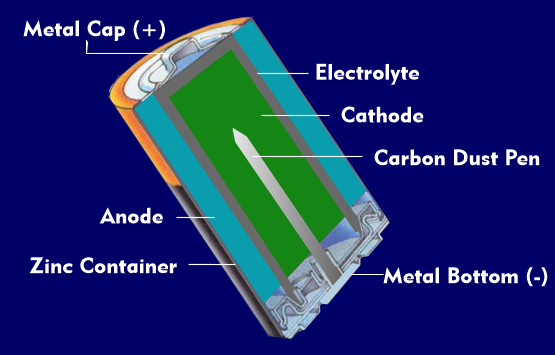
battery cell
A battery cell is a single electricity-generating unit that stores chemical energy and converts it into electrical energy. Several battery cells connected together form a battery. The battery voltage is the sum of the battery cell voltages.
A battery cell has two electrodes, the cathode and the anode, and an electrolyte, which is a chemical substance that reacts with the electrodes to produce electrical current. Such a battery cell is also called a galvanic element. If a battery cell cannot be charged, it is a primary cell, if it can be recharged after discharge, it is a secondary cell, an accumulator.
The nominal voltage of a battery cell depends on the electrode material and results from the electrochemical voltage series. Depending on the material, the voltage values range between +2.9 V and -3.0 V.